Table Of Content
In an internal pilot study, the first phase of the study is designated a “pilot phase,” and the study is continued till this sample size is achieved (definitive phase) and analysis incorporates the pilot subjects also. In contrast to external pilots, internal pilots can be large, as they do not “use up” eligible patients and do not require additional time or funds. The three most common types of endpoints in clinical trials are continuous endpoints (e.g., pain on a visual analogue scale), categorical (including binary, e.g., response vs. no response) endpoints, and event-time endpoints (e.g., time to death).
Publication types
Key opinion leaders on GLP-1 receptor agonist clinical trial design - Clinical Trials Arena
Key opinion leaders on GLP-1 receptor agonist clinical trial design.
Posted: Fri, 26 Apr 2024 15:02:48 GMT [source]
A common design feature is the use of central labs for quantitating laboratory parameters to eliminate between-lab variation or the use of central evaluators to eliminate between-evaluator variation. For example, the AIDS Clinical Trials Group (ACTG) uses a central laboratory to quantitate HIV-1 RNA viral load on all of its studies while trials using imaging modalities for diagnose stroke might consider using a central imaging laboratory to quantitate all images. In this section, we outline the different areas that need careful attention when considering a clinical trial. Is an unpaid member of the board of directors of Quantum Leap Healthcare Collaborative (QLHC, the study sponsor), and received grant funding from QLHC for the I-SPY TRIAL. Is a member of the Blue Cross/Blue Shield Medical Advisory Panel and receives reimbursement for time and travel.
Overview of clinical trial phases
For example, the quantity to be estimated may be the between-group difference in the mean response. A sample size is then calculated to ensure that there is a high probability that this quantity is estimated with acceptable precision as measured by say the width of the confidence interval for the between-group difference in means. In selecting a population to enroll into a trial, researchers must consider the target use of the intervention since it will be desirable to generalize the results of the trial to the target population. However researchers also select entry criteria to help ensure a high quality trial and to address the specific objectives of the trial.
Clinical research study designs: The essentials
It comprises two single-vector compounds with arenaviral backbones based on lymphocytic choriomeningitis virus (LCMV) and pichinde virus (PICV). Both express the same transgene encoding an E7E6 fusion protein derived from HPV16. HB-200 is an alternating 2-vector immunotherapy designed to further focus the immune response against the encoded antigen. Assessing the risk/benefit ratio does not mean to automatically reject high risk or low benefit proposals, but rather to adjust the design to minimize cost to subjects and society. In this module, you’ll learn how to ethically and effectively recruit and retain the participants you need for your trial and strategically select the clinical sites where you’ll conduct your research.
Complex Innovative Trial Design Meeting Program - FDA.gov
Complex Innovative Trial Design Meeting Program.
Posted: Thu, 11 Apr 2024 07:00:00 GMT [source]
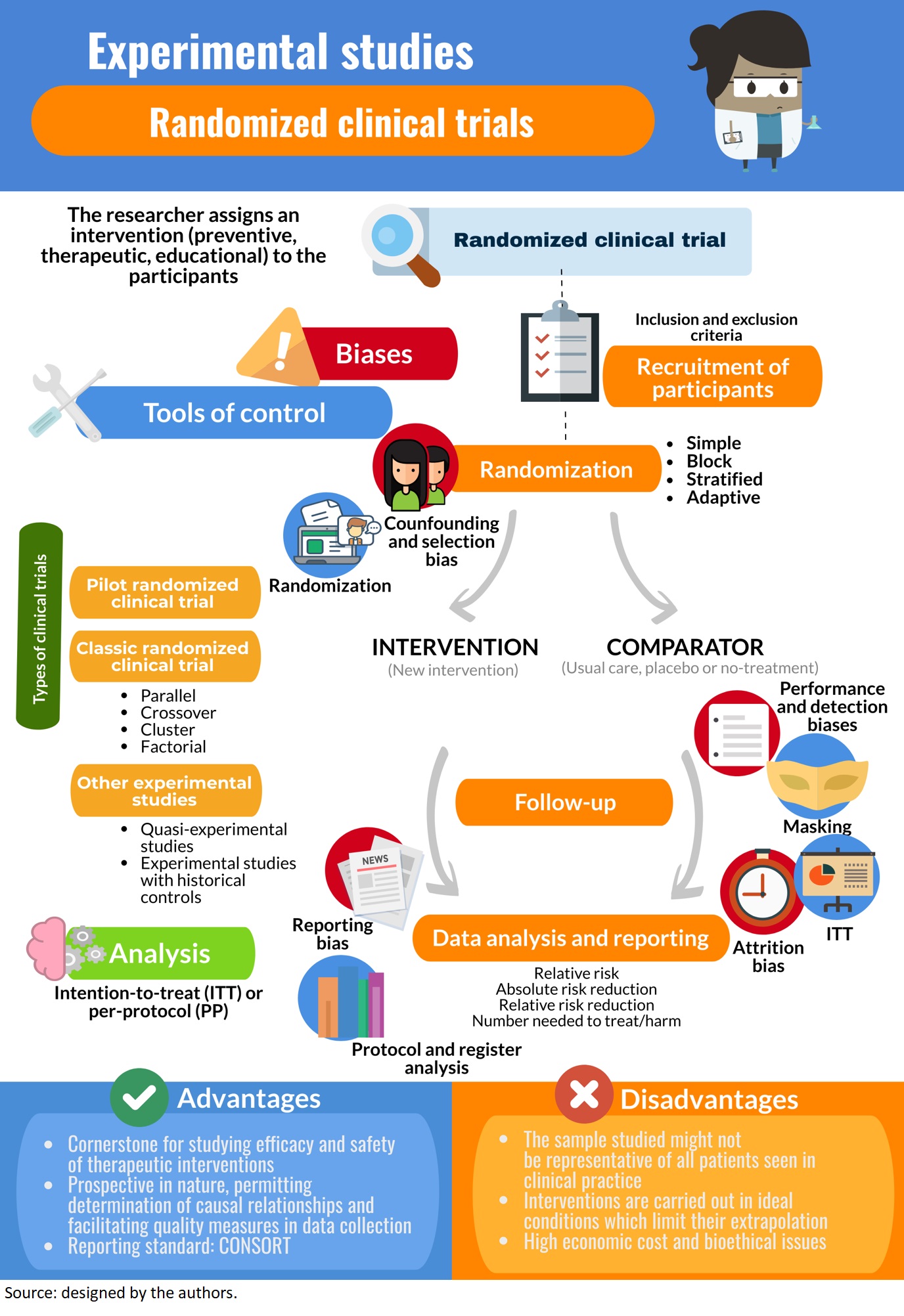
This week focuses on a key issue in the field of clinical trials, the ethics of experimentation in humans. In block randomization, the subjects of similar characteristics are classified into blocks. The aim of block randomization is to balance the number of subjects allocated to each experiment/intervention group. For example, let's assume that there are four subjects in each block, and two of the four subjects in each block will be randomly allotted to each group. Therefore, there will be two subjects in one group and two subjects in the other group.17 The disadvantage with this methodology is that there is still a component of predictability in the selection of subjects and the randomization of prognostic factors is not performed.
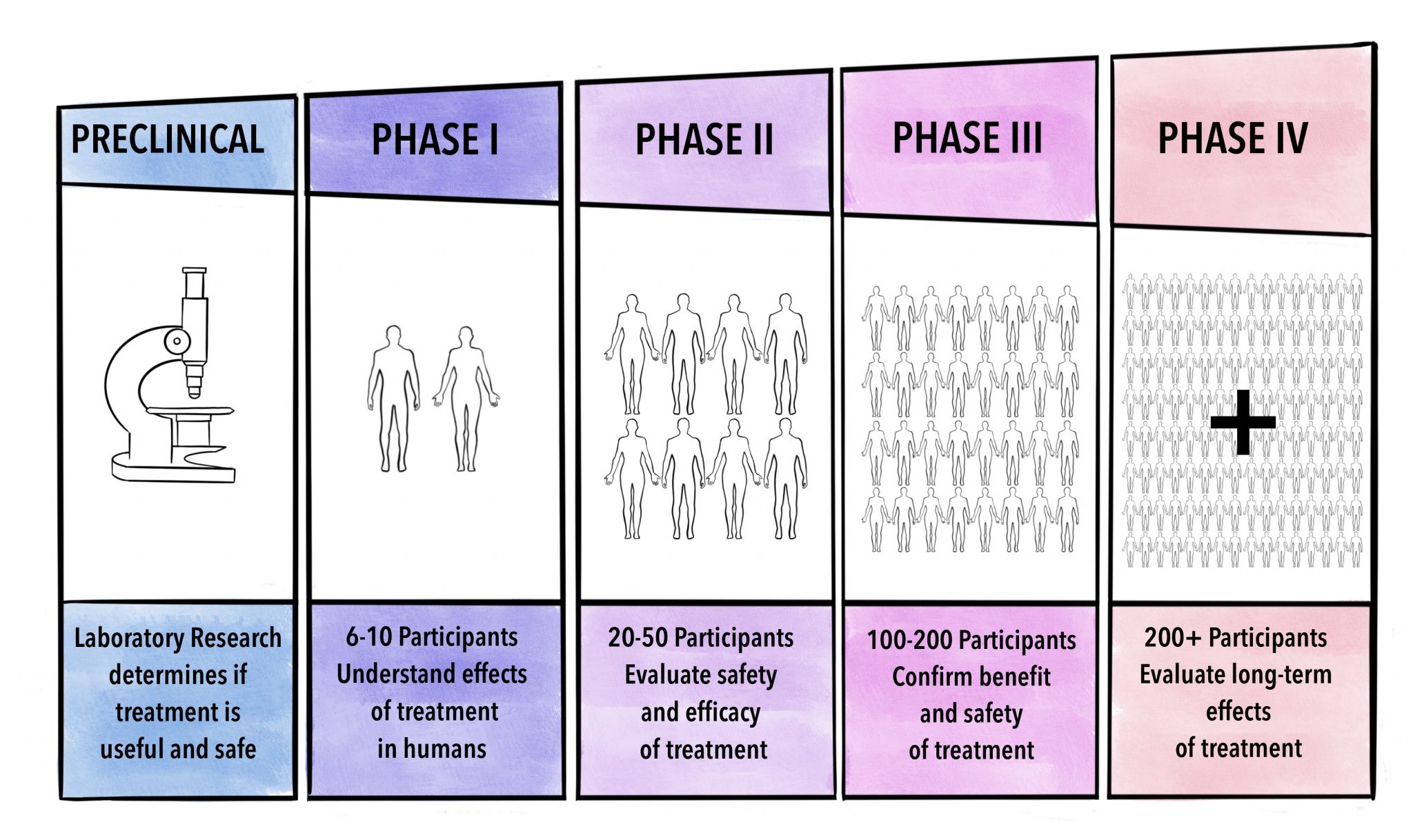
Knowledge gaps should not be filled with assumptions – if data is lacking, the hypothesis and research question should be broken down to something simpler that can be supported and answered within a clinical trial. It is important that the investigator clearly understands the stage of development of the drug or device being tested. Phases of development are classic for drug trials, and are described as phases I-IV as we explain below.
Clinical trial phases: pharmacological studies
Meanwhile, blinding protects against ascertainment bias (changing the outcome on knowing what intervention was received). Unblinded assessors might score outcomes differently; unblinded subjects may report symptoms differently, become less adherent, or drop out at higher rates (if they get sham treatment). However, not all studies can be blinded feasibly or ethically; e.g. many surgical interventions cannot be blinded for these reasons. We highlight the need for investigators to incorporate best practice in trial design to increase the chances of success, to always anticipate unexpected challenges during the trial. Second, cooperation across a wide variety of stakeholders was essential for both spurring innovation and speeding implementation.
Selection of Trial Designs
It is not meant to be a strict set of rules to be followed in some prescribed order, rather it is meant to be a set of guidelines to consider in active collaboration with the study team including a statistician. These principles should apply for designing any clinical trial, regardless of who initiates and conducts the study (e.g. research group vs. industry). The involvement of the statistician throughout the entire research cannot be overemphasized. The statistician can aid in each step, from formulating appropriate scientific hypotheses to designing and conducting simulation studies. In addition to being collaborative, the design process is also iterative; it may be that some design elements need to be modified after other design elements are considered. For example, trial phase is typically driven by the level of available evidence on the drug being tested.
Cohort studies are typically chosen as a study design when the suspected exposure is known and rare, and the incidence of disease/outcome in the exposure group is suspected to be high. The choice between prospective and retrospective cohort study design would depend on the accuracy and reliability of the past records regarding the exposure/risk factor. Every now and then during clinical practice, we come across a case that is atypical or ‘out of the norm’ type of clinical presentation.
A factorial trial study design is adopted when the researcher wishes to test two different drugs with independent effects on the same population. Typically, the population is divided into 4 groups, the first with drug A, the second with drug B, the third with drug A and B, and the fourth with neither drug A nor drug B. The outcomes for drug A are compared to those on drug A, drug A and B and to those who were on drug B and neither drug A nor drug B.15 The advantages of this study design that it saves time and helps to study two different drugs on the same study population at the same time. However, this study design would not be applicable if either of the drugs or interventions overlaps with each other on modes of action or effects, as the results obtained would not attribute to a particular drug or intervention. The experimental study design can be classified into 2 groups, that is, controlled (with comparison) and uncontrolled (without comparison).1 In the group without controls, the outcome is directly attributed to the treatment received in one group. This fails to prove if the outcome was truly due to the intervention implemented or due to chance.
Although clinical trials are conducted prospectively, one can think of them as being designed retrospectively. That is, there is a vision of the scientific claim (i.e., answer to the research question) that a project team would like to make at the end of the trial. In order to make that claim, appropriate analyses must be conducted in order to justify the claim.

No comments:
Post a Comment